|
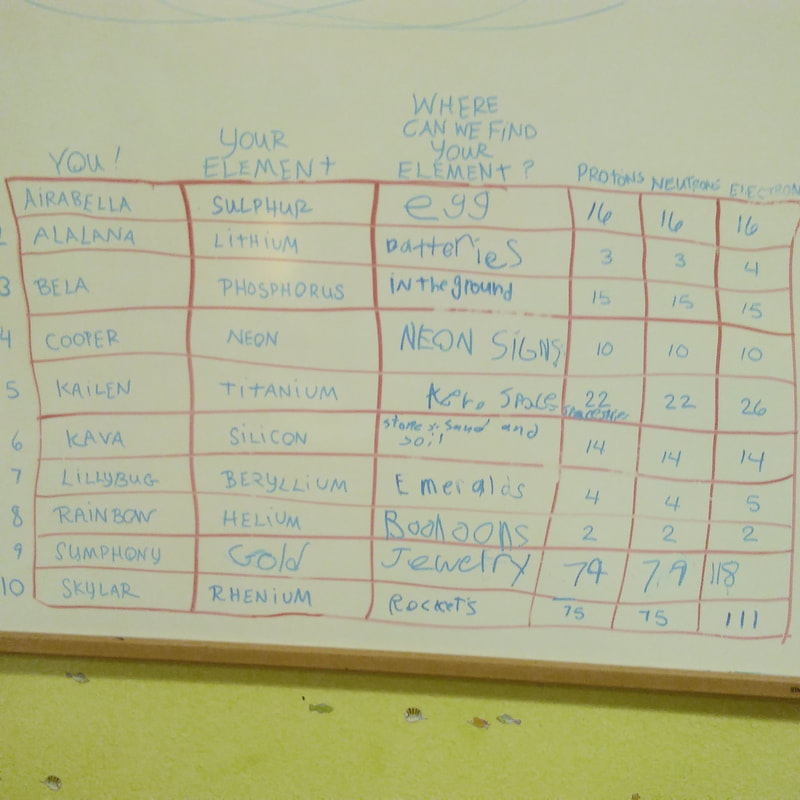
It all started last spring, when I drank too much coffee after school, found an exciting Periodic Table of the Elements curriculum, and decided to print ALL 80 PAGES. No matter that our curriculum didn't plan for Chemistry last spring, nor that we'd already booked ourselves with TMTDBSES (Too Much To Do Before School Ends Syndrome). I printed those pages anyway, hole-punched them into a binder, then waited for the molecule's moment. Months went by. The new year began, and within two weeks we'd already reached critical TMTDBSES mode. Yet in October --- suddenly!! --- we were gifted with an open morning and science to teach. Well, why not now? First, each child chose an element: Then we created atomic structures from clay. Inside the nucleus of each atom are protons (with a positive electrical charge) and neutrons (with no electrical charge), and the kids picked two different colors to paint their protons and neutrons: Our atomic nuclei will stay in the classroom, yet the electrons are experiencing a very different story! For, you see, we learned through our friend Bill Nye (the Science Guy) that if electrons were actually the size of our clay balls, they would actually be 500 meters away from the nucleus! (So, even though atoms and mostly made up of EMPTY SPACE between the nucleus and electrons, they give the appearance of being solid because the electrons are moving so, so, so, so fast around the atom --kind of like a ceiling fan that behaves like a solid when it moves quickly). And because we are realistic-minded folks, we took a walk with our meter tool: But we reached the end of the soccer field after only 150 meters! We "planted" our electrons ... ...with promises to return to walk the entire 500 meters (which will lead us from school grounds and into the Takilma community!).
Comments are closed.
|

 RSS Feed
RSS Feed
